Catalysts | Free Full-Text | Palladium-Based Catalysts as Electrodes for Direct Methanol Fuel Cells: A Last Ten Years Review

Self-assembled palladium nanoflowers supported on fullerene: Electrochemical catalytic performance for the reduction of 4-nitrophenol - ScienceDirect

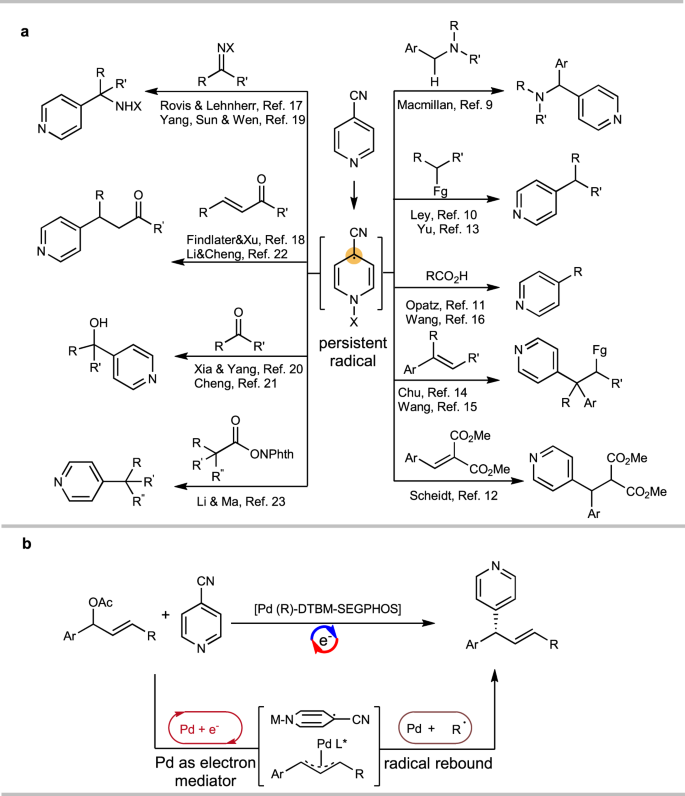
On the Mechanism of Palladium-Catalyzed Aromatic C−H Oxidation | Journal of the American Chemical Society
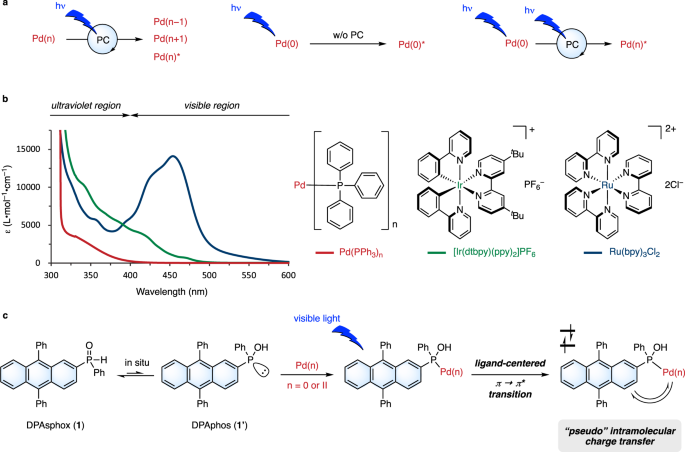
A visible-light activated secondary phosphine oxide ligand enabling Pd-catalyzed radical cross-couplings | Nature Communications

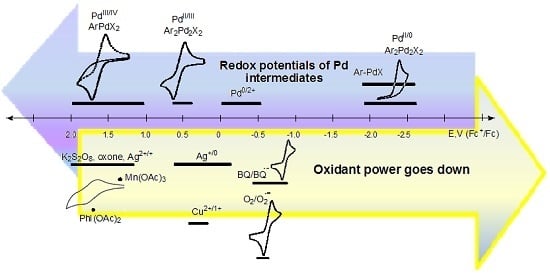
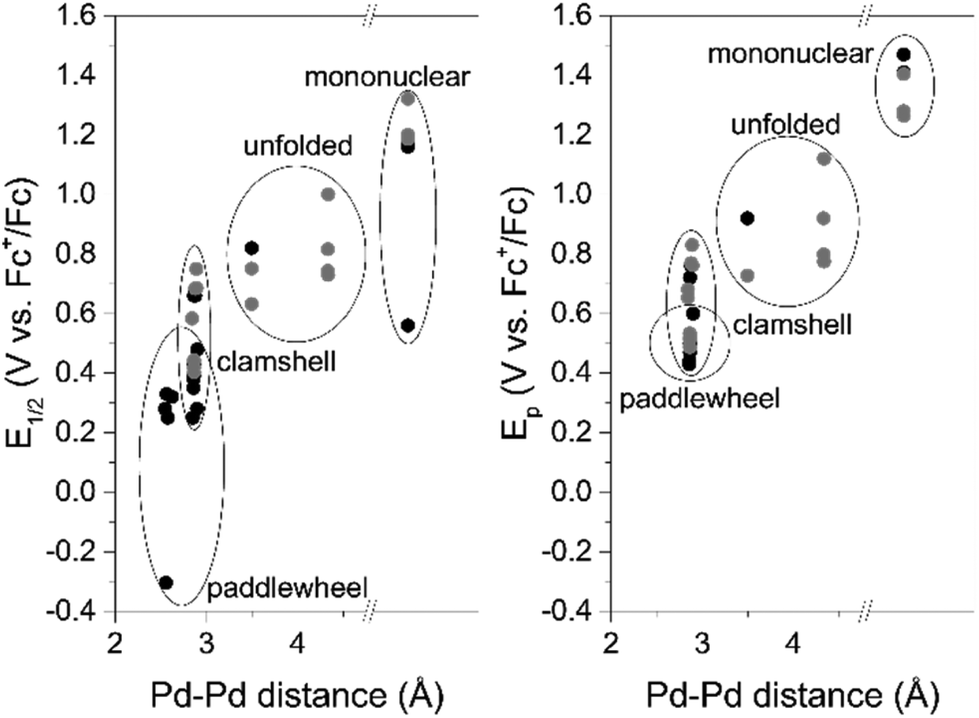
Redox trends in cyclometalated palladium( ii ) complexes - Dalton Transactions (RSC Publishing) DOI:10.1039/C6DT03786K
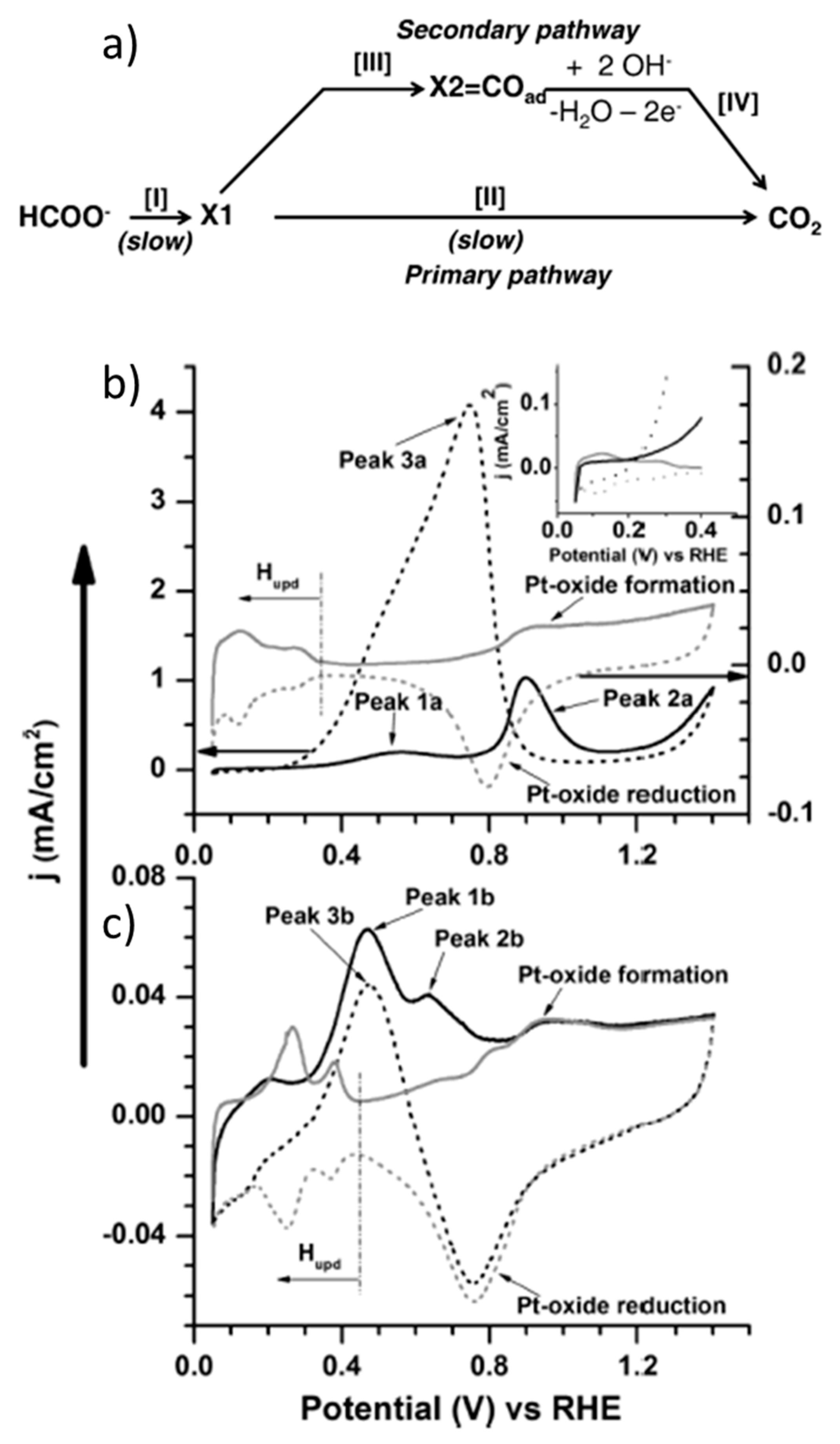
Inorganics | Free Full-Text | Redox-Induced Aromatic C–H Bond Functionalization in Metal Complex Catalysis from the Electrochemical Point of View

Can Donor Ligands Make Pd(OAc)2 a Stronger Oxidant? Access to Elusive Palladium(II) Reduction Potentials and Effects of Ancillary Ligands via Palladium(II)/Hydroquinone Redox Equilibria | Journal of the American Chemical Society

Tuning the Copper(II)/Copper(I) Redox Potential for More Robust Copper‐Catalyzed C–N Bond Forming Reactions - Cope - 2020 - European Journal of Inorganic Chemistry - Wiley Online Library

Can Donor Ligands Make Pd(OAc)2 a Stronger Oxidant? Access to Elusive Palladium(II) Reduction Potentials and Effects of Ancillary Ligands via Palladium(II)/Hydroquinone Redox Equilibria | Journal of the American Chemical Society

Solvent-Induced Reduction of Palladium-Aryls, a Potential Interference in Pd Catalysis | Organometallics
Redox trends in cyclometalated palladium( ii ) complexes - Dalton Transactions (RSC Publishing) DOI:10.1039/C6DT03786K