Clinical Outcomes and Adverse Events after First-Line Treatment in Metastatic Renal Cell Carcinoma: A Systematic Review and Netw
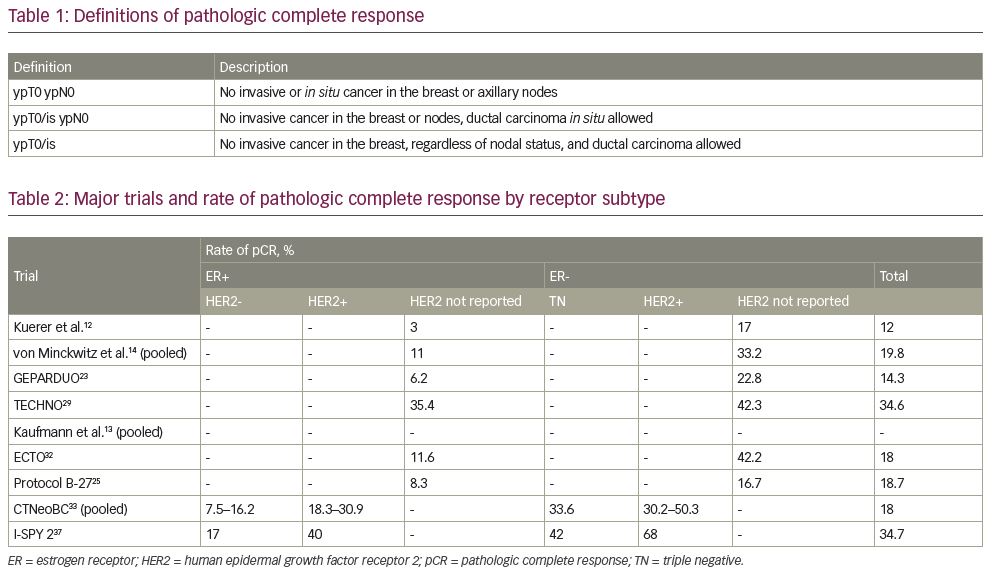
Physical exercise during neoadjuvant chemotherapy for breast cancer as a mean to increase pathological complete response rates: Trial protocol of the randomized Neo-ACT trial | PLOS ONE

Abbreviations: CR, complete response; ORR, objective response rate; PR,... | Download Scientific Diagram

Response rates Overall response rate and depth of response according to... | Download Scientific Diagram
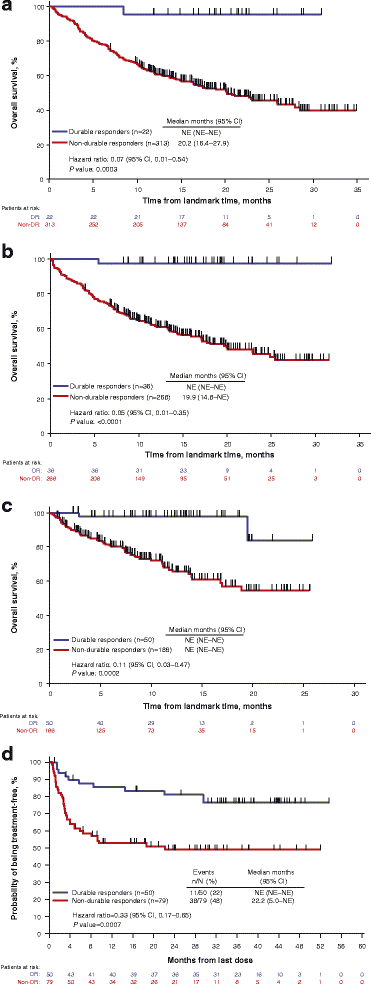
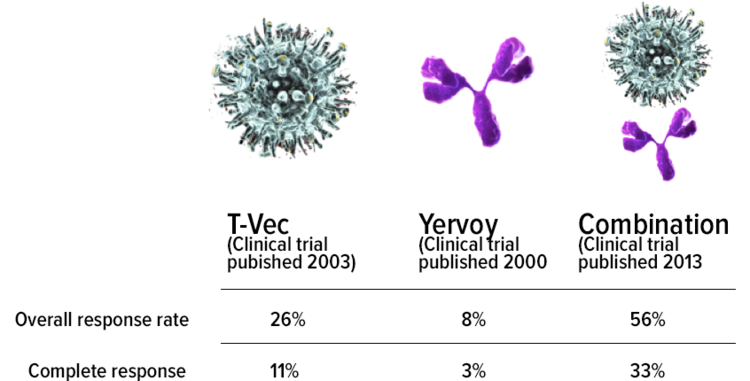
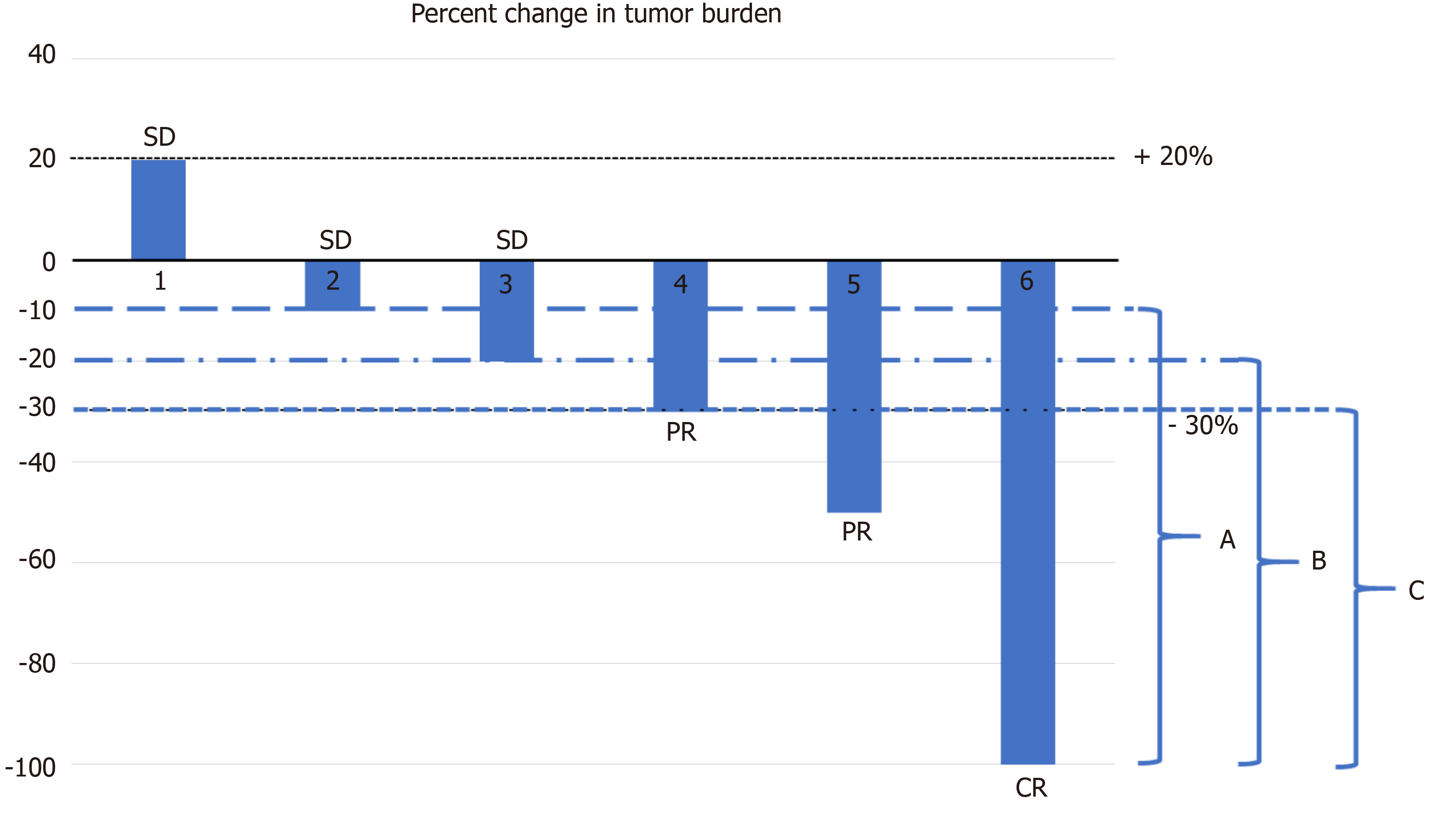
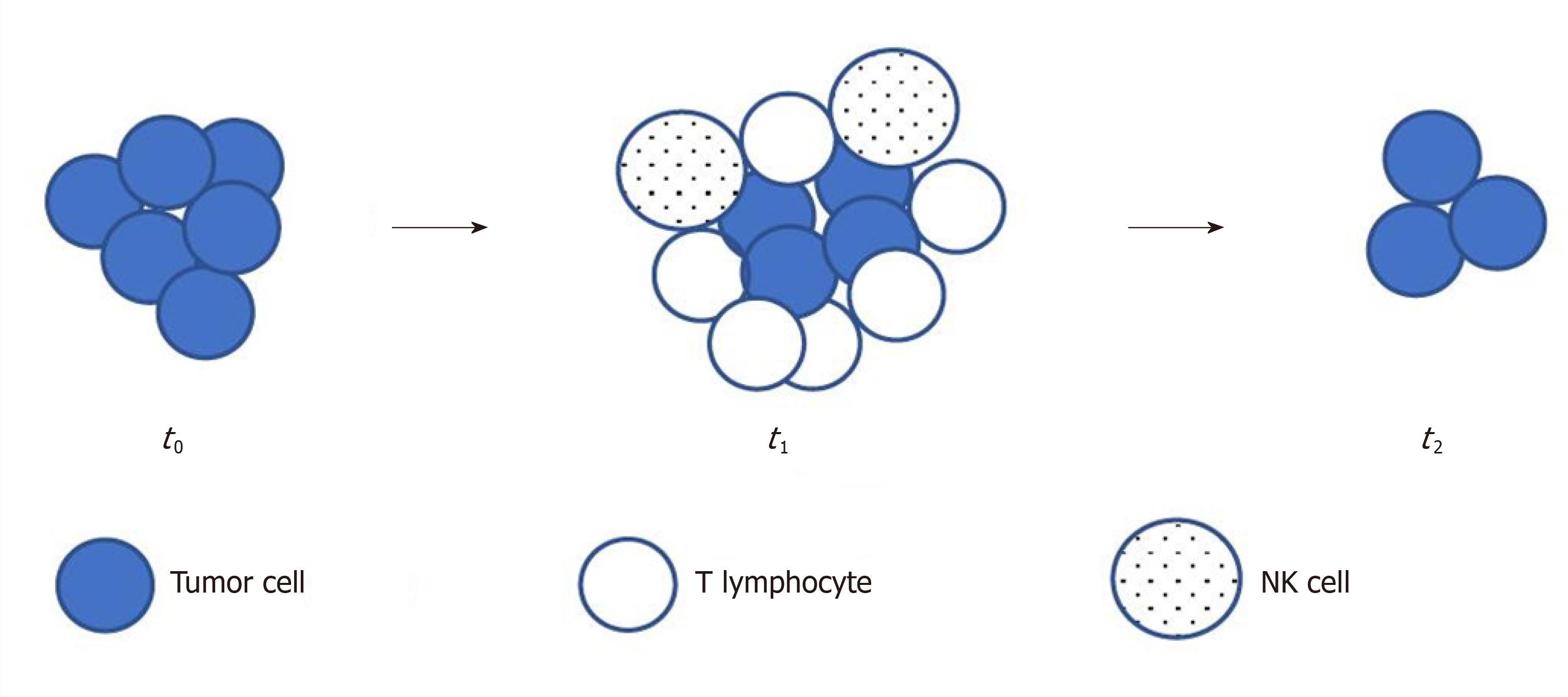
Durable response rate as an endpoint in cancer immunotherapy: insights from oncolytic virus clinical trials | Journal for ImmunoTherapy of Cancer | Full Text
Physical exercise during neoadjuvant chemotherapy for breast cancer as a mean to increase pathological complete response rates: Trial protocol of the randomized Neo-ACT trial | PLOS ONE

Relationship Between Progression‐Free Survival, Objective Response Rate, and Overall Survival in Clinical Trials of PD‐1/PD‐L1 Immune Checkpoint Blockade: A Meta‐Analysis - Ye - 2020 - Clinical Pharmacology & Therapeutics - Wiley Online
KAZIA PRESENTS FURTHER PAXALISIB AND CANTRIXIL DATA AT AACR, REINFORCING POSITIVE EFFICACY SIGNALS FOR BOTH DRUGS