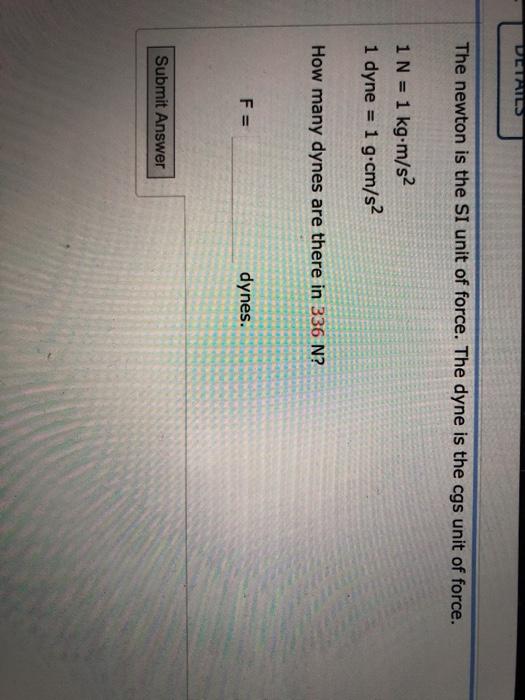
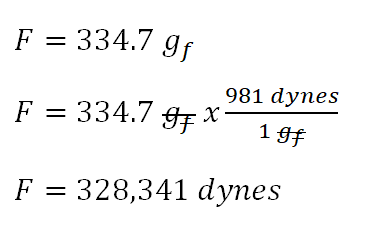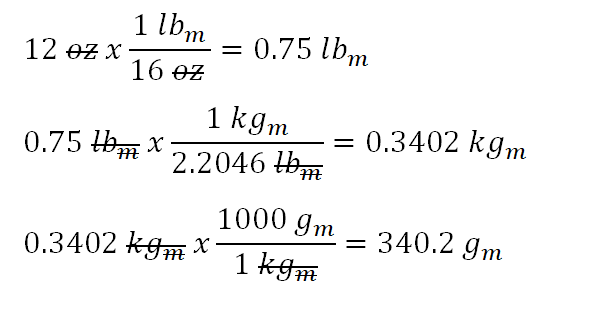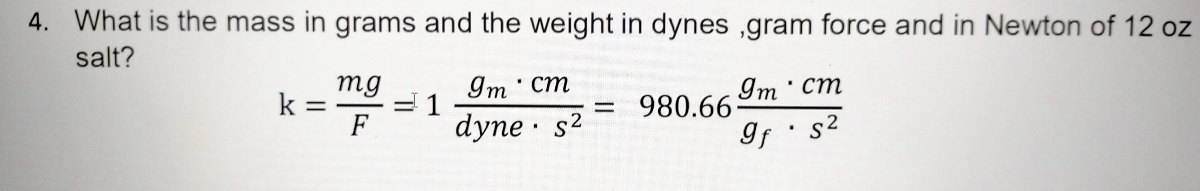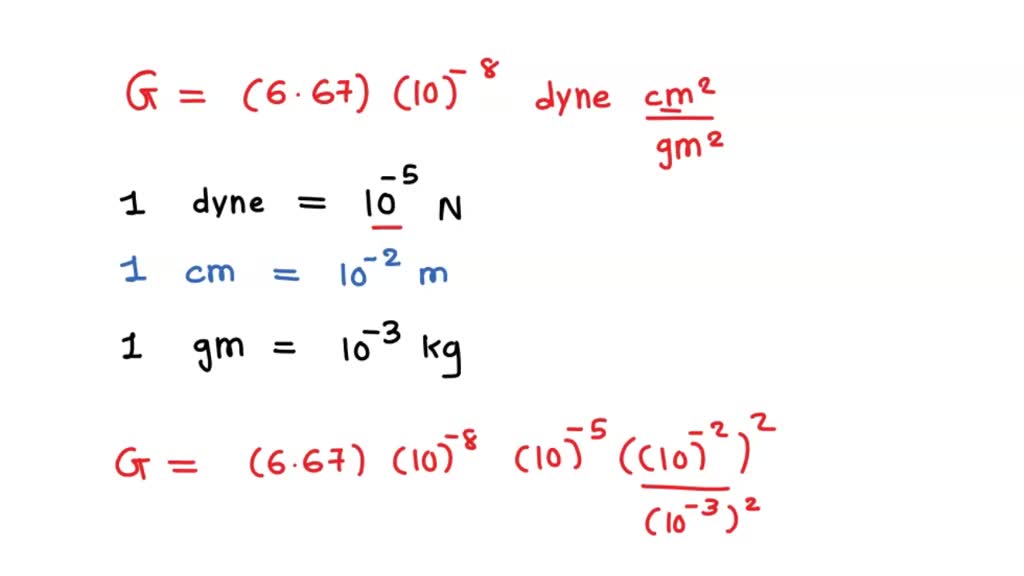Using Dimensional Analysis to Find the Quantity of a Substance Practice | Chemistry Practice Problems | Study.com

Activity 1: solve for size and speed of a drop.. Useful units for calculations Remember: dyne = 1 g cm/s 2 (force in cm-g-s units) = Newtons. Using. - ppt download

Dyne/square centimeter to Gram-force/sq. centimeter Converter: 1 Dyne/square centimeter in Gram-force/sq. centimeter
The value of Gravitional cons†an t in MKS system is 6.67×10^ 11 N m^2/kg^2. What will be its value in CGS system