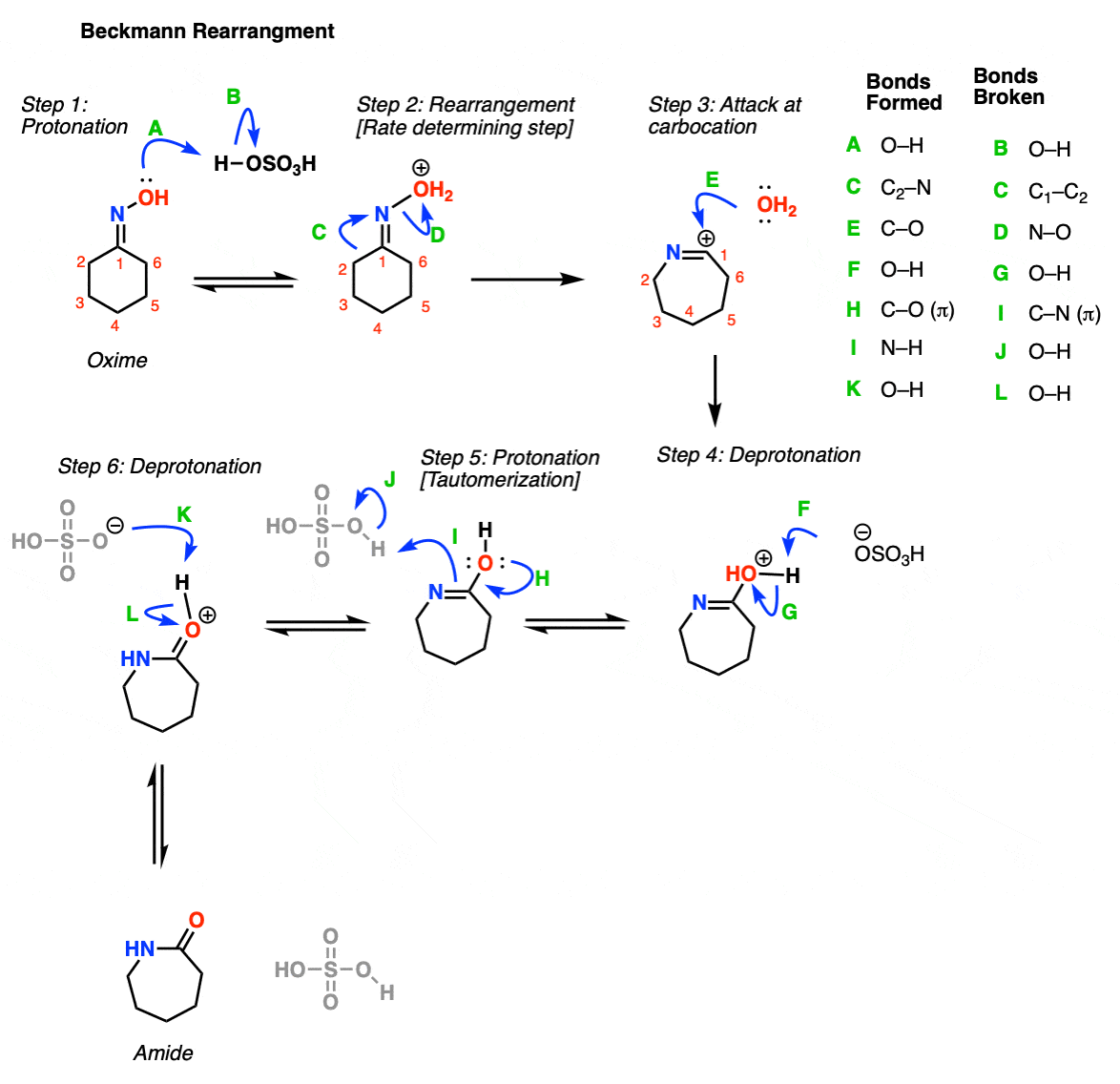
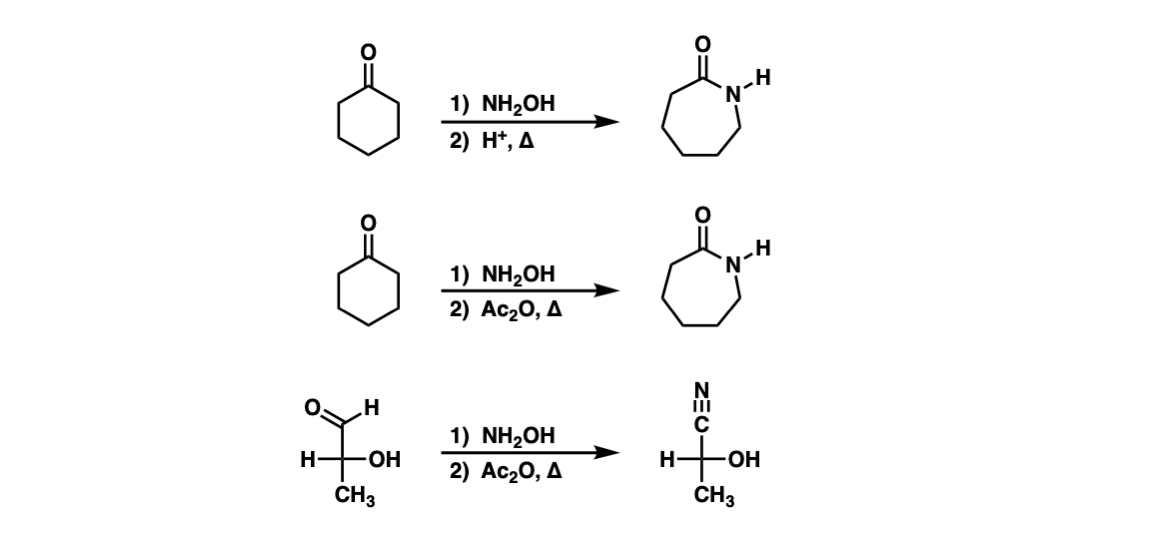
Photocatalyzed Triplet Sensitization of Oximes Using Visible Light Provides a Route to Nonclassical Beckmann Rearrangement Products | Journal of the American Chemical Society

An Electrochemical Beckmann Rearrangement: Traditional Reaction via Modern Radical Mechanism - Tang - 2020 - ChemSusChem - Wiley Online Library

CXVIII.—The oxidation of d-Δ4-carene with Beckmann's chromic acid mixture - Journal of the Chemical Society (Resumed) (RSC Publishing)

Dichloroimidazolidinedione-Activated Beckmann Rearrangement of Ketoximes for Accessing Amides and Lactams | The Journal of Organic Chemistry

Oxidation Leading to Reduction: Redox‐Induced Electron Transfer (RIET) - Miller - 2009 - Angewandte Chemie International Edition - Wiley Online Library
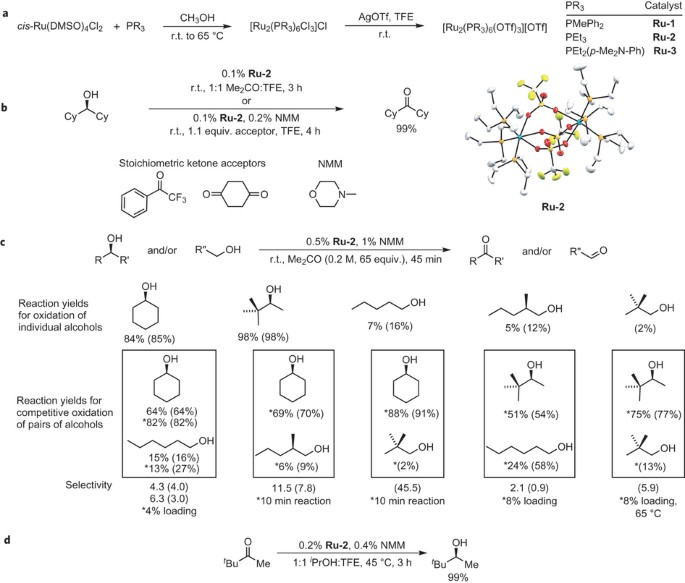
Site-selective oxidation, amination and epimerization reactions of complex polyols enabled by transfer hydrogenation | Nature Chemistry

A Stable Primary Phosphane Oxide and Its Heavier Congeners - Horký - 2021 - Chemistry – A European Journal - Wiley Online Library

Marine Drugs | Free Full-Text | Structure Elucidation of Calyxoside B, a Bipolar Sphingolipid from a Marine Sponge Cladocroce sp. through the Use of Beckmann Rearrangement

Silica-Supported TEMPO Catalysts: Synthesis and Application in the Anelli Oxidation of Alcohols | The Journal of Organic Chemistry

Visible Light‐Promoted Beckmann Rearrangements: Separating Sequential Photochemical and Thermal Phenomena in a Continuous Flow Reactor - Chen - 2019 - European Journal of Organic Chemistry - Wiley Online Library
![SOLVED: Analysis 5: Compare the stepwise mechanism of Schmidt reaction (II) with that of Beckmann rearrangement; then with that of Baeyer-Villiger oxidation. HN-OH MCPBA pTSA HN] Analysis 6: Design synthetic route for SOLVED: Analysis 5: Compare the stepwise mechanism of Schmidt reaction (II) with that of Beckmann rearrangement; then with that of Baeyer-Villiger oxidation. HN-OH MCPBA pTSA HN] Analysis 6: Design synthetic route for](https://cdn.numerade.com/ask_images/8d7a3665a3a640ceb573f2f6fd0bd525.jpg)
SOLVED: Analysis 5: Compare the stepwise mechanism of Schmidt reaction (II) with that of Beckmann rearrangement; then with that of Baeyer-Villiger oxidation. HN-OH MCPBA pTSA HN] Analysis 6: Design synthetic route for





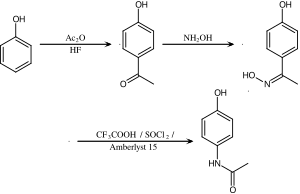

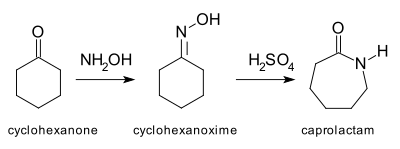

![Nitrosation and Beckmann reactions. Modified from ref. [6]. | Download Scientific Diagram Nitrosation and Beckmann reactions. Modified from ref. [6]. | Download Scientific Diagram](https://www.researchgate.net/publication/303086293/figure/fig1/AS:361640055853059@1463233116334/Nitrosation-and-Beckmann-reactions-Modified-from-ref-6.png)